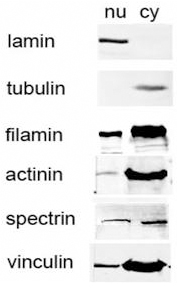
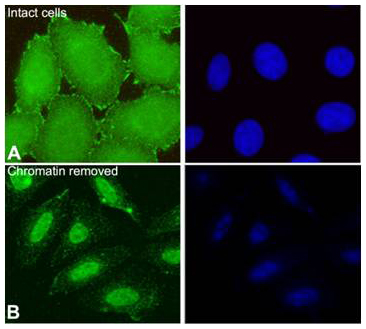
| Introduction |
|
In diploid mammalian
cells, some 6x109 base pairs of DNA
fold as a nucleo-protein complex
(i.e. chromatin) into higher-order arrays so as to fit in a nucleus
measuring
only 10 µm. The nucleus also contains machineries for
transcription of genes
and processing of RNA products, and for precise DNA replication, repair
and
recombination. Nuclear interior is therefore functionally highly
compartmentalized, and the recent evidence points strongly to
structure-related
regulation of nuclear functions – however, the mechanisms forming the
3D-structure of the nucleus are still mostly obscure. We therefore
employ a
multi-disciplinary approach in order to study nuclear functions in
relation to
the higher-order nuclear structures, e.g. nuclear bodies, the
nucleolus, and
the nucleoskeleton. Our research concentrates on:
|